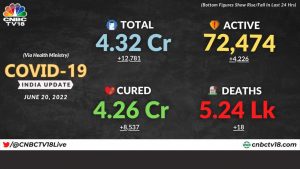
One-third of participants who took Covaxin report adverse events, says new study
Summary
The study, conducted by Sankha Shubhra Chakrabarti and his team at Banaras Hindu University, found that nearly a third of participants reported adverse events of special interest (aesi) among the participants who took Covaxin.
A significant rate of adverse events of special interest (AESI) was reported among the participants of Bharat Biotech’s Covid-19 vaccine, Covaxin, a new study has revealed.
The study found that nearly one-third of the participants experienced AESI, according to a report on SpringerLink, an integrated platform for journals and other materials published by Springer.
It is also revealed that female adolescents and individuals with a history of allergies are at a higher risk of AESI.
The study was conducted by Sankha Shubhra Chakrabarti and his team at Banaras Hindu University. She said, “The majority of the AESI persisted at the one-year follow-up.”
During the one-year follow-up, 635 adolescents and 291 adults of the 1,024 enrolled persons were contacted. The study further revealed that 124 (42.6%) adults and 304 (47.9%) adolescents reported having viral upper respiratory tract infections after taking the vaccine.
The most common AESIs in adolescents were new-onset skin and subcutaneous disorders (10.5%), general disorders (10.2%), and nervous system disorders (4.7%).
Meanwhile, in adults, the common AESIs reported were nervous system disorders (5.5%), musculoskeletal disorders (5.8%), and general disorders (8.9%).
Additional reported issues were menstrual abnormalities in 4.6% of female participants, ocular abnormalities in 2.7%, and hypothyroidism in 0.6% of participants.
Serious AESIs such as stroke and Guillain-Barre Syndrome were noticed in 0.3% and 0.1% of participants, respectively.
The study also said that the participants with a history of allergy and post-vaccination typhoid were at 1.6, 2.8, and 2.8 times higher risk of AESIs, and the majority of AESIs occur during one-year follow-up.
Females and adolescents with a history of pre-vaccination COVID-19, comorbidities, or post-vaccination typhoid had 1.6, 2, 2.7, and 3.2 times higher odds of persistent AESIs. Adults with comorbidities had more than twice the risk of both AESIs and persistent AESIs compared to others.
Adolescents and adults experienced different patterns of adverse effects after being administered Covaxin than were observed with other COVID-19 vaccinations.
The researchers also emphasised the necessity of extended surveillance of COVID-19-vaccinated persons to understand the long-term effects and outcomes of these adverse events.
“Focused monitoring for persistent AESIs is warranted for individuals with a pre-vaccination history of COVID,” the researchers said.
“Adults with comorbidities, such as hypertension, are at a higher risk of AESIs and persistent AESIs after BBV152 administration. The patterns of AESIs developing after Covaxin differ from those seen after Covishield, as well as among different age groups,” it added.

Elon Musk forms several ‘X Holdings’ companies to fund potential Twitter buyout
3 Mins Read
Thursday’s filing dispelled some doubts, though Musk still has work to do. He and his advisers will spend the coming days vetting potential investors for the equity portion of his offer, according to people familiar with the matter









 Listen to the Article
Listen to the Article  Daily Newsletter
Daily Newsletter