USFDA issues inspection report for Lupin’s Aurangabad manufacturing facility
Summary
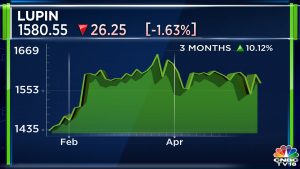
Shares of Lupin Ltd ended at ₹1,580.55, down by ₹26.25, or 1.63% on the BSE.
Drug maker Lupin Ltd on Tuesday (April 23) said it has received the Establishment Inspection Report (EIR) from the United States Food and Drug Administration (US FDA) for its Aurangabad manufacturing facility.
The inspection was conducted from March 6 to March 15, 2024. The US FDA has determined that the inspection classification of the facility is Voluntary Action Indicated (VAI), Lupin said in a regulatory filing.
Also Read: Nelco declares dividend of ₹2.20 per share, Q4 net profit up 7% to ₹6 crore
Nilesh Gupta, managing director of Lupin, said, “We are pleased to receive the EIR with VAI status from the US FDA as an outcome of the recent inspection of our Aurangabad facility.
It is a testament to our commitment to consistently upholding the highest standards of compliance and providing high-quality healthcare solutions to patients worldwide.”
Shares of Lupin Ltd ended at ₹1,580.55, down by ₹26.25, or 1.63% on the BSE.

Elon Musk forms several ‘X Holdings’ companies to fund potential Twitter buyout
3 Mins Read
Thursday’s filing dispelled some doubts, though Musk still has work to do. He and his advisers will spend the coming days vetting potential investors for the equity portion of his offer, according to people familiar with the matter








 Listen to the Article
Listen to the Article  Daily Newsletter
Daily Newsletter




